Abstract
Introduction: Ixazomib is an oral proteasome inhibitor (PI) that is currently approved to be administered once weekly in combination with lenalidomide (LEN) and dexamethasone in RRMM (Moreau et al N Eng J Med 2016; 374:1621-1634). As most patients are LEN refractory at the time of first relapse, pomalidomide-based regimens are commonly utilized due to their proven efficacy in this population. We hypothesized that twice weekly dosing of ixazomib may be more efficacious as this has been previously studied as monotherapy (Richardson et al, Blood 2014 Aug 14;124(7):1038-46) and in combination with LEN demonstrating promising activity and safety (Richardson et al, Br J Haematol. 2018 Jul;182(2):231-244). We present results of our phase I/II trial of twice weekly ixazomib in combination with pomalidomide and dexamethasone in RRMM, including the recommended phase II dose and first report of efficacy of this combination.
Methods: This is a phase I/II multicenter, single-arm, open label study evaluating the combination of twice weekly ixazomib with pomalidomide and dexamethasone in RRMM. The primary objective for phase I portion is to determine safety and the maximum tolerated dose (MTD) of this combination using a standard 3+3 dose escalation design and primary objective of the phase II portion is overall response rate (ORR) with secondary outcomes including progression-free survival (PFS) and clinical benefit rate (CBR)
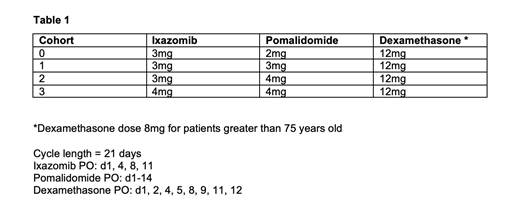
Ixazomib is studied at doses of 3mg or 4mg on days 1, 4, 8, 11, pomalidomide at a dose of 2mg, 3mg and 4mg on days 1-14 and dexamethasone is administered at a dose of 12mg on days 1, 2, 4, 5, 8, 9, 11, 12 (8mg for patients > 75 years old) on a 21 day cycle (Table 1). Patients were included if they received 2 prior lines of therapy, but 1 prior line was allowed if first line treatment included a PI and an immunomodulatory agent and disease relapse occurred within 60 days of last therapy. Patients who were ixazomib exposed or pomalidomide refractory were excluded.
Results: At the time of data cutoff, 22 patients have been enrolled across all cohorts. There were two dose-limiting toxicities (DLT) noted during the dose escalation phase (upper respiratory infection and neutropenia, respectively) establishing the RP2D of 4mg ixazomib and 4mg pomalidomide. Median age at the time of enrollment was 68 years old with ISS stage at diagnosis of I (14%), II (32%), and III (23%). High-risk FISH abnormalities were seen in 43% of patients as follows: del 17p (9%), gain 1q (36%), t(4;14) (5%), t(14;16) (9%). Median prior lines of therapy was 2 (range 1-4) with 100% of patients having prior treatment with lenalidomide and 95% with prior bortezomib. Fifty-nine percent of patients had a prior autologous stem cell transplant. Ten patients have been enrolled at the RP2D at the time of data cut off.
The most common treatment-related toxicities were mainly low grade (Grade 1-2) and included neutropenia (45%), lower extremity edema (41%), insomnia (36%), dyspnea (32%) and weight gain (32%). Grade 3 or greater toxicities were noted in 36% of patients and included neutropenia (18%), thrombocytopenia (5%), anemia (5%), atrial fibrillation (5%), dehydration (5%), diarrhea (5%), fall (5%), lung infection (5%), and pneumonitis (5%). Dose reductions occurred in 13 patients and predominantly involved dexamethasone due to weight gain, insomnia, atrial fibrillation and fatigue. There have been no discontinuations due to toxicity and no treatment related mortality at the time of data cutoff.
The ORR in all cohorts was 45%, with 9% achieving sCR, 9% VGPR and 86% achieving stable disease or better. At the RP2D, the ORR was 50% with 30% of patients achieving VGPR or better. At the median follow up of 10 months, median PFS was 13 months (95% CI: 11-NR) and median overall survival was not reached.
Conclusions: Twice weekly ixazomib in combination with pomalidomide and dexamethasone is a generally well-tolerated regimen with promising activity. The recommended phase II dose has been established at 4mg of ixazomib and 4mg of pomalidomide demonstrating efficacy in a high-risk cohort of RRMM patients. The all-oral nature of this regimen has allowed for robust accrual during the COVID 19 pandemic.
Nadeem: BMS: Membership on an entity's Board of Directors or advisory committees; Karyopharm: Membership on an entity's Board of Directors or advisory committees; GSK: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Adaptive Biotechnologies: Membership on an entity's Board of Directors or advisory committees. Mo: Karyopharm: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria; GSK: Consultancy, Membership on an entity's Board of Directors or advisory committees; Epizyme: Consultancy; Eli Lilly: Consultancy; BMS: Membership on an entity's Board of Directors or advisory committees; Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees; AbbVIE: Consultancy. Bianchi: Jacob D. Fuchsberg Law Firm: Consultancy; MJH: Honoraria; Karyopharm: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria. Sanchorawala: Celgene: Research Funding; Pfizer: Honoraria; Sorrento: Research Funding; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Research Funding; Prothena: Membership on an entity's Board of Directors or advisory committees, Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees; Caelum: Membership on an entity's Board of Directors or advisory committees, Research Funding; Regeneron: Membership on an entity's Board of Directors or advisory committees; Proclara: Membership on an entity's Board of Directors or advisory committees; Oncopeptide: Research Funding; Karyopharm: Research Funding. Sperling: Adaptive: Consultancy. Munshi: Janssen: Consultancy; Takeda: Consultancy; Bristol-Myers Squibb: Consultancy; Celgene: Consultancy; Amgen: Consultancy; Karyopharm: Consultancy; Oncopep: Consultancy, Current equity holder in publicly-traded company, Other: scientific founder, Patents & Royalties; Legend: Consultancy; Abbvie: Consultancy; Adaptive Biotechnology: Consultancy; Novartis: Consultancy; Pfizer: Consultancy. Ghobrial: AbbVie, Adaptive, Aptitude Health, BMS, Cellectar, Curio Science, Genetch, Janssen, Janssen Central American and Caribbean, Karyopharm, Medscape, Oncopeptides, Sanofi, Takeda, The Binding Site, GNS, GSK: Consultancy. Anderson: Sanofi-Aventis: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Gilead: Membership on an entity's Board of Directors or advisory committees; Millenium-Takeda: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees; AstraZeneca: Membership on an entity's Board of Directors or advisory committees; Scientific Founder of Oncopep and C4 Therapeutics: Current equity holder in publicly-traded company, Current holder of individual stocks in a privately-held company; Mana Therapeutics: Membership on an entity's Board of Directors or advisory committees. Richardson: AbbVie: Consultancy; Karyopharm: Consultancy, Research Funding; Janssen: Consultancy; Protocol Intelligence: Consultancy; Takeda: Consultancy, Research Funding; GlaxoSmithKline: Consultancy; Regeneron: Consultancy; Secura Bio: Consultancy; Sanofi: Consultancy; AstraZeneca: Consultancy; Celgene/BMS: Consultancy, Research Funding; Oncopeptides: Consultancy, Research Funding; Jazz Pharmaceuticals: Consultancy, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal